
July 2021. Article published in PNAS: Research Sheds Light on Forests’ Role in Transferring the Atmospheric Pollutant to the Environment
By Edwin L. Aguirre. Toxic mercury is being deposited in forests in much higher quantities than previously thought, posing a concern for the health and well-being of people, wildlife and waterways, according to a UMass Lowell scientist investigating the source of the pollution. A team of researchers led by Prof. Daniel Obrist, chairman of the Department of Environmental, Earth and Atmospheric Sciences, conducted comprehensive measurement of mercury accumulation in a forest, including deposition of the gaseous form of the element. Forests constitute the world’s most abundant, productive and widespread land ecosystems.
“It’s the first such study that covers, at the ecosystem level, the full picture of mercury deposition at any forest site in the world,” says Obrist. “Plants take up gaseous mercury from the atmosphere through their leaves, through their stomata (pores), and as the plants shed their leaves or die off, they basically transfer that atmospheric mercury to the ecosystems. Mercury deposits in forests ultimately run off into streams and rivers, ending up in lakes and oceans,” he explains.
The team’s data, which was gathered at a forest in north-central Massachusetts, showed the pronounced and dominant role of gaseous mercury for annual deposition, amounting to 25 micrograms of mercury per square meter of forest. This is five times greater than mercury deposited by rain and snow, according to Obrist. “Our observed gaseous deposition accounts for 76% of the total mercury deposition from the atmosphere. It’s also three times greater than mercury deposition through litterfall (dead leaves that fall to the ground in autumn) alone, which has previously been used as a proxy for estimating gaseous mercury deposition in forests,” he notes.
The results of the project, which is supported by a three-year, $873,000 grant from the National Science Foundation (NSF), will be published this July in an issue of the Proceedings of the National Academy of Sciences. Other contributors to the study include Asst. Prof. Róisín Commane of Columbia University; students and postdoctoral researchers from UMass Lowell and Columbia University; and collaborators from Harvard University; the Desert Research Institute in Reno, Nevada; and the Northwest Institute of Eco-Environment and Resources and the University of the Chinese Academy of Sciences in Lanzhou. Additional research support was provided by the U.S. Department of Energy.
Link to the article:
https://www.pnas.org/content/118/29/e2105477118.short
Links to media articles:
https://www.uml.edu/News/stories/2021/Obrist-PNAS-paper.aspx
http://news.climate.columbia.edu/2021/07/12/study-shows-forests-may-take-in-far-more-toxic-mercury-than-thought/
https://www.nsf.gov/discoveries/disc_summ.jsp?cntn_id=303188&org=NSF&from=news
By Edwin L. Aguirre. Toxic mercury is being deposited in forests in much higher quantities than previously thought, posing a concern for the health and well-being of people, wildlife and waterways, according to a UMass Lowell scientist investigating the source of the pollution. A team of researchers led by Prof. Daniel Obrist, chairman of the Department of Environmental, Earth and Atmospheric Sciences, conducted comprehensive measurement of mercury accumulation in a forest, including deposition of the gaseous form of the element. Forests constitute the world’s most abundant, productive and widespread land ecosystems.
“It’s the first such study that covers, at the ecosystem level, the full picture of mercury deposition at any forest site in the world,” says Obrist. “Plants take up gaseous mercury from the atmosphere through their leaves, through their stomata (pores), and as the plants shed their leaves or die off, they basically transfer that atmospheric mercury to the ecosystems. Mercury deposits in forests ultimately run off into streams and rivers, ending up in lakes and oceans,” he explains.
The team’s data, which was gathered at a forest in north-central Massachusetts, showed the pronounced and dominant role of gaseous mercury for annual deposition, amounting to 25 micrograms of mercury per square meter of forest. This is five times greater than mercury deposited by rain and snow, according to Obrist. “Our observed gaseous deposition accounts for 76% of the total mercury deposition from the atmosphere. It’s also three times greater than mercury deposition through litterfall (dead leaves that fall to the ground in autumn) alone, which has previously been used as a proxy for estimating gaseous mercury deposition in forests,” he notes.
The results of the project, which is supported by a three-year, $873,000 grant from the National Science Foundation (NSF), will be published this July in an issue of the Proceedings of the National Academy of Sciences. Other contributors to the study include Asst. Prof. Róisín Commane of Columbia University; students and postdoctoral researchers from UMass Lowell and Columbia University; and collaborators from Harvard University; the Desert Research Institute in Reno, Nevada; and the Northwest Institute of Eco-Environment and Resources and the University of the Chinese Academy of Sciences in Lanzhou. Additional research support was provided by the U.S. Department of Energy.
Link to the article:
https://www.pnas.org/content/118/29/e2105477118.short
Links to media articles:
https://www.uml.edu/News/stories/2021/Obrist-PNAS-paper.aspx
http://news.climate.columbia.edu/2021/07/12/study-shows-forests-may-take-in-far-more-toxic-mercury-than-thought/
https://www.nsf.gov/discoveries/disc_summ.jsp?cntn_id=303188&org=NSF&from=news

April 2021. Article published in Nature Review - Earth and Environment
By Nancy Cicco and Christine Gillette.
LOWELL, Mass. – Researchers studying mercury gas in the atmosphere with the aim of reducing the pollutant worldwide have determined a vast amount of the toxic element is absorbed by plants, leading it to deposit into soils.
Hundreds of tons of mercury each year are emitted into the atmosphere as a gas by burning coal, mining and other industrial and natural processes. These emissions are absorbed by plants in a process similar to how they take up carbon dioxide. When the plants shed leaves or die, the mercury is transferred to soils where large amounts also make their way into watersheds, threatening wildlife and people who eat contaminated fish. Exposure to high levels of mercury over long periods can lead to neurological and cardiovascular problems in humans, according to UMass Lowell’s Daniel Obrist, professor and chair of the Department of Environmental, Earth and Atmospheric Sciences, who is leading the research group.
Obrist is an expert on the cycling of mercury in the environment. In his latest project, he and UMass Lowell Research Associate Jun Zhou collected more than 200 published studies with data on mercury levels in vegetation from more than 400 locations around the world. In evaluating this data, they determined about 88 percent of the mercury found in plants originates from plants’ leaves absorbing gaseous mercury from the atmosphere. Globally, vegetation can take up more than 1,300 tons of mercury each year, accounting for 60 to 90 percent of it being deposited over land, according to Zhou. The team’s findings were published this month in the academic journal Nature Reviews – Earth & Environment. The study represents the largest comprehensive review of the uptake of mercury in vegetation and its impact on mercury cycling around the world, according to the researchers.
“When I walk outside here in New England, I am always amazed at the greenness of our forest, grasslands and salt marshes. One goal of my research is to determine how strongly vegetation controls the cycling of elements – some of which can be toxic pollutants – so we can better mitigate damaging effects,” Obrist said. The work moves scientists toward a greater understanding of how mercury cycling works, according to Zhou. “Researchers have worked on the role that vegetation plays on cycling of mercury for over 30 years now, but the full extent of these impacts are still not yet fully realized. It was timely to write this comprehensive review and communicate to colleagues and the public about the current state of knowledge in this area,” Zhou said.
Other contributors to the study include scientists from the Environment and Climate Change Canada’s Air Quality Research Division in Quebec, and the University of Basel in Switzerland. Support for the research was provided by the U.S. National Science Foundation and Swiss National Science Foundation.
Link to the article:
https://rdcu.be/cgQ14
Links to media articles:
https://www.uml.edu/News/press-releases/2021/ObristReviewRelease033121.aspx
https://scienmag.com/researchers-plants-play-leading-role-in-cycling-toxic-mercury-through-the-environment/
https://phys.org/news/2021-03-role-toxic-mercury-environment.html
By Nancy Cicco and Christine Gillette.
LOWELL, Mass. – Researchers studying mercury gas in the atmosphere with the aim of reducing the pollutant worldwide have determined a vast amount of the toxic element is absorbed by plants, leading it to deposit into soils.
Hundreds of tons of mercury each year are emitted into the atmosphere as a gas by burning coal, mining and other industrial and natural processes. These emissions are absorbed by plants in a process similar to how they take up carbon dioxide. When the plants shed leaves or die, the mercury is transferred to soils where large amounts also make their way into watersheds, threatening wildlife and people who eat contaminated fish. Exposure to high levels of mercury over long periods can lead to neurological and cardiovascular problems in humans, according to UMass Lowell’s Daniel Obrist, professor and chair of the Department of Environmental, Earth and Atmospheric Sciences, who is leading the research group.
Obrist is an expert on the cycling of mercury in the environment. In his latest project, he and UMass Lowell Research Associate Jun Zhou collected more than 200 published studies with data on mercury levels in vegetation from more than 400 locations around the world. In evaluating this data, they determined about 88 percent of the mercury found in plants originates from plants’ leaves absorbing gaseous mercury from the atmosphere. Globally, vegetation can take up more than 1,300 tons of mercury each year, accounting for 60 to 90 percent of it being deposited over land, according to Zhou. The team’s findings were published this month in the academic journal Nature Reviews – Earth & Environment. The study represents the largest comprehensive review of the uptake of mercury in vegetation and its impact on mercury cycling around the world, according to the researchers.
“When I walk outside here in New England, I am always amazed at the greenness of our forest, grasslands and salt marshes. One goal of my research is to determine how strongly vegetation controls the cycling of elements – some of which can be toxic pollutants – so we can better mitigate damaging effects,” Obrist said. The work moves scientists toward a greater understanding of how mercury cycling works, according to Zhou. “Researchers have worked on the role that vegetation plays on cycling of mercury for over 30 years now, but the full extent of these impacts are still not yet fully realized. It was timely to write this comprehensive review and communicate to colleagues and the public about the current state of knowledge in this area,” Zhou said.
Other contributors to the study include scientists from the Environment and Climate Change Canada’s Air Quality Research Division in Quebec, and the University of Basel in Switzerland. Support for the research was provided by the U.S. National Science Foundation and Swiss National Science Foundation.
Link to the article:
https://rdcu.be/cgQ14
Links to media articles:
https://www.uml.edu/News/press-releases/2021/ObristReviewRelease033121.aspx
https://scienmag.com/researchers-plants-play-leading-role-in-cycling-toxic-mercury-through-the-environment/
https://phys.org/news/2021-03-role-toxic-mercury-environment.html

March 2021. New site at Howland Forest instrumented to measure gaseous mercury deposition in forests
By Jun Zhou: Studies suggest that 60% to 90% of Hg in forest ecosystems originate from vegetation uptake of atmospheric gaseous elemental Hg(0), providing about 50% of total global terrestrial deposition of 1,310 to 1,570 Mg yr-1. Forest are known as important mercury deposition sinks and act as a global atmospheric Hg(0) pump. However, the magnitude and controls of Hg(0) deposition over evergreen needle forests in northern areas are not known.
Our group, in collaboration with the University of Maine, the U.S. Forest Service, and Columbia University, is measuring the forest’s uptake of atmospheric Hg(0) at Howland Forest in Maine, an evergreen needle forest dominated by red spruce and eastern hemlock. We installed analyzers and automated sensors to measure gaseous Hg(0) on existing large towers at various heights from above the tree canopy down to near the forest floor. Additionally, we are also measuring other trace gases, such as carbon dioxide, ozone, methane, water vapor, and carbonyl sulfide, which will allow us to assess the mechanisms of mercury uptake by plants and underlying soils. The results of these observations in the northern needle forest will help to reduce uncertainties in global atmospheric mercury deposition rates and residence times across forests.
By Jun Zhou: Studies suggest that 60% to 90% of Hg in forest ecosystems originate from vegetation uptake of atmospheric gaseous elemental Hg(0), providing about 50% of total global terrestrial deposition of 1,310 to 1,570 Mg yr-1. Forest are known as important mercury deposition sinks and act as a global atmospheric Hg(0) pump. However, the magnitude and controls of Hg(0) deposition over evergreen needle forests in northern areas are not known.
Our group, in collaboration with the University of Maine, the U.S. Forest Service, and Columbia University, is measuring the forest’s uptake of atmospheric Hg(0) at Howland Forest in Maine, an evergreen needle forest dominated by red spruce and eastern hemlock. We installed analyzers and automated sensors to measure gaseous Hg(0) on existing large towers at various heights from above the tree canopy down to near the forest floor. Additionally, we are also measuring other trace gases, such as carbon dioxide, ozone, methane, water vapor, and carbonyl sulfide, which will allow us to assess the mechanisms of mercury uptake by plants and underlying soils. The results of these observations in the northern needle forest will help to reduce uncertainties in global atmospheric mercury deposition rates and residence times across forests.

June 2020. New study funded by the National Science Foundation Division of Environmental Biology
By Daniel Obrist: The goal of our new project is to determine the origin and fate of mercury in a salt marsh ecosystem in the Plum Island Sound of Massachusetts. Along the U.S. East Coast, salt marsh sparrows have shown harmful exposures to mercury with elevated blood levels posing risks for reproductive outputs. Possible reasons for high mercury exposure are contributions from interior watersheds or legacy industrial impacts, yet the origin of mercury in these salt marshes is largely unknown. This project will study the role of plant uptake of atmospheric mercury by salt marsh plants as possible dominant source for these ecosystems, analogue to observations from forests that show atmospheric mercury uptake by plants to constitute up to 90% of mercury inputs. In this tidal system, an additional focus lies on the transfer of mercury to soils and its export to the coastal ocean. The project will involve graduate students and provide experiential learning opportunities for undergraduate students.
Funding source: U.S. NSF DEB #2027038: Collaborative Research: Vegetation assimilation as a source of mercury in a salt marsh ecosystem and implications for soil and tidal water exposures. Obrist D (PI), Forbrich I (Co-PI).
$399,208 (UMass component: 298,242).
By Daniel Obrist: The goal of our new project is to determine the origin and fate of mercury in a salt marsh ecosystem in the Plum Island Sound of Massachusetts. Along the U.S. East Coast, salt marsh sparrows have shown harmful exposures to mercury with elevated blood levels posing risks for reproductive outputs. Possible reasons for high mercury exposure are contributions from interior watersheds or legacy industrial impacts, yet the origin of mercury in these salt marshes is largely unknown. This project will study the role of plant uptake of atmospheric mercury by salt marsh plants as possible dominant source for these ecosystems, analogue to observations from forests that show atmospheric mercury uptake by plants to constitute up to 90% of mercury inputs. In this tidal system, an additional focus lies on the transfer of mercury to soils and its export to the coastal ocean. The project will involve graduate students and provide experiential learning opportunities for undergraduate students.
Funding source: U.S. NSF DEB #2027038: Collaborative Research: Vegetation assimilation as a source of mercury in a salt marsh ecosystem and implications for soil and tidal water exposures. Obrist D (PI), Forbrich I (Co-PI).
$399,208 (UMass component: 298,242).
New Graduate Student in the Graduate Program of Environmental Studies at UMass Lowell.
By Christ Romero. I am a first year graduate student working towards my M.S in Environmental Studies with a concentration in Geosciences at The University of Massachusetts at Lowell, where I joined the Obrist Lab group in the fall of 2019. I hold a B.S in Geology from The University of Massachusetts at Amherst and have previously conducted direct oceanographic research on ocean acidification during my time at Sea Education Association/Sea Semester, where my passion for climate change research began. My research interest includes atmospheric mercury pollution and climate change influences on the environment. I am currently working on Harvard Forest Gaseous Elemental Mercury Hg(0) Flux project, based out of Petersham, MA, USA. My research focuses on atmospheric exchange fluxes of Hg(0) at the ecosystem-level along with observed atmospheric Hg(0) concentrations in order to understand the dynamics of Hg(0) at the canopy and forest floor levels of a temperate deciduous broadleaf forest. This is possible by implementing flux gradient methods along with micrometeorological measuring techniques. My research includes field work such as the servicing/maintenance of field instruments, quality control and data science management by employing the statistical modeling software STATA along with programing language Python/packages and. The exchange of Hg(0) between atmospheric, terrestrial and aquatic ecosystem compartments is of great concern, due to the production of the toxin substance MeHg, where it can strongly bioaccumulates through the food chain posing a risk to wild life and human health.
By Christ Romero. I am a first year graduate student working towards my M.S in Environmental Studies with a concentration in Geosciences at The University of Massachusetts at Lowell, where I joined the Obrist Lab group in the fall of 2019. I hold a B.S in Geology from The University of Massachusetts at Amherst and have previously conducted direct oceanographic research on ocean acidification during my time at Sea Education Association/Sea Semester, where my passion for climate change research began. My research interest includes atmospheric mercury pollution and climate change influences on the environment. I am currently working on Harvard Forest Gaseous Elemental Mercury Hg(0) Flux project, based out of Petersham, MA, USA. My research focuses on atmospheric exchange fluxes of Hg(0) at the ecosystem-level along with observed atmospheric Hg(0) concentrations in order to understand the dynamics of Hg(0) at the canopy and forest floor levels of a temperate deciduous broadleaf forest. This is possible by implementing flux gradient methods along with micrometeorological measuring techniques. My research includes field work such as the servicing/maintenance of field instruments, quality control and data science management by employing the statistical modeling software STATA along with programing language Python/packages and. The exchange of Hg(0) between atmospheric, terrestrial and aquatic ecosystem compartments is of great concern, due to the production of the toxin substance MeHg, where it can strongly bioaccumulates through the food chain posing a risk to wild life and human health.

Immersive Scholar joined our group.
By Sam Charron: I am currently a Freshman at UMass Lowell and a member of the Honors College Immersive Scholars Program. Via the Immersive Scholars program, I have had the opportunity to join a team of researchers and an ongoing study to assess mercury (Hg) deposition at Harvard Forest. During my time in the research team, I have produced and analyzed graphs illustrating diurnal and seasonal trends of various atmospheric trace gases (carbon dioxide, Hg) and wind/pollution roses depicting how weather patterns influence Hg pollution patterns. Apart from learning more about Hg cycling, I have also learned to work with graphing and statistical programs such as Stata and Python, how to code within the program Anaconda, and have made large improvements in my scientific writing.
By Sam Charron: I am currently a Freshman at UMass Lowell and a member of the Honors College Immersive Scholars Program. Via the Immersive Scholars program, I have had the opportunity to join a team of researchers and an ongoing study to assess mercury (Hg) deposition at Harvard Forest. During my time in the research team, I have produced and analyzed graphs illustrating diurnal and seasonal trends of various atmospheric trace gases (carbon dioxide, Hg) and wind/pollution roses depicting how weather patterns influence Hg pollution patterns. Apart from learning more about Hg cycling, I have also learned to work with graphing and statistical programs such as Stata and Python, how to code within the program Anaconda, and have made large improvements in my scientific writing.
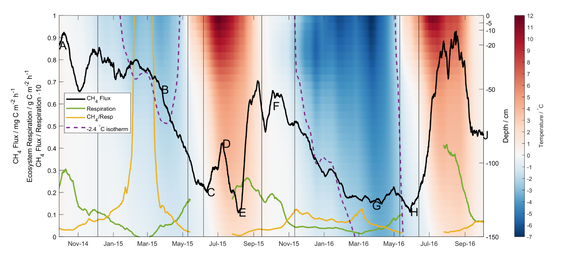
July 2020: Paper on arctic methane fluxes published in the journal Biogeosciences
By Daniel Obrist: Congratulations to Dean Howard, former postdoc in our group and now at the University of Colorado, Boulder, for leading a new paper published in the journal Biogeosciences. Harsh wintertime conditions have limited the number of year-round ecosystem-level methane and carbon dioxide flux studies in the Arctic. This study presents a two-year micrometeorological flux records of methane and carbon dioxide that provide near-continuous data including wintertime data in the tundra of northern Alaska. Net emissions of methane and carbon dioxide in one of the study years totalled 3.7 and 89 g C m−2 a−1 respectively, with cold season methane emission representing 54% of the annual total. In the other year, net emission totals of methane and carbon dioxide were 4.9 and 485 g C m−2 a−1 respectively, with cold season methane emission representing 82% of the annual total. This study shows that cold-season methane fluxes dominated as source of methane at this Arctic tundra site. We propose that deep soil horizons (~1 m) where most microbial methanogenic activity takes place remained warm enough to maintain efficient methane production throughout winter, whilst surface soil temperatures were simultaneously cold enough to limit methanotrophic activity (methane consumption).
Full citation: Howard D, Agnan Y, Helmig D, Yang Y, Obrist D. Environmental controls on ecosystem-scale cold season methane and carbon dioxide fluxes in an Arctic tundra ecosystem. Biogeosciences, in press.
By Daniel Obrist: Congratulations to Dean Howard, former postdoc in our group and now at the University of Colorado, Boulder, for leading a new paper published in the journal Biogeosciences. Harsh wintertime conditions have limited the number of year-round ecosystem-level methane and carbon dioxide flux studies in the Arctic. This study presents a two-year micrometeorological flux records of methane and carbon dioxide that provide near-continuous data including wintertime data in the tundra of northern Alaska. Net emissions of methane and carbon dioxide in one of the study years totalled 3.7 and 89 g C m−2 a−1 respectively, with cold season methane emission representing 54% of the annual total. In the other year, net emission totals of methane and carbon dioxide were 4.9 and 485 g C m−2 a−1 respectively, with cold season methane emission representing 82% of the annual total. This study shows that cold-season methane fluxes dominated as source of methane at this Arctic tundra site. We propose that deep soil horizons (~1 m) where most microbial methanogenic activity takes place remained warm enough to maintain efficient methane production throughout winter, whilst surface soil temperatures were simultaneously cold enough to limit methanotrophic activity (methane consumption).
Full citation: Howard D, Agnan Y, Helmig D, Yang Y, Obrist D. Environmental controls on ecosystem-scale cold season methane and carbon dioxide fluxes in an Arctic tundra ecosystem. Biogeosciences, in press.

November 2019. New postodoctoral fellow, Jun Zhou, joined our group
By Jun Zhou: I joined the Obrist lab in November 2019 after working as assistant researcher in the Institute of Soil Science, at the Chinese Academy of Sciences. My research interests include the study of heavy metal (mainly mercury) fluxes between terrestrial ecosystem and atmosphere and their effects on surface environments and soils. Currently, I am working largely on two reserach projects: (i) investigation of gaseous atmospheric mercury deposition in forests and (ii) assessing sources of mercury in a salt marsh ecosystem. My research encompasses both field and laboratory work, including plant assimilation of atmospheric mercury, surface-air mercury fluxes, atmospheric and soil mercury speciation, and the dynamics and fate of mercury in soils after deposition.
By Jun Zhou: I joined the Obrist lab in November 2019 after working as assistant researcher in the Institute of Soil Science, at the Chinese Academy of Sciences. My research interests include the study of heavy metal (mainly mercury) fluxes between terrestrial ecosystem and atmosphere and their effects on surface environments and soils. Currently, I am working largely on two reserach projects: (i) investigation of gaseous atmospheric mercury deposition in forests and (ii) assessing sources of mercury in a salt marsh ecosystem. My research encompasses both field and laboratory work, including plant assimilation of atmospheric mercury, surface-air mercury fluxes, atmospheric and soil mercury speciation, and the dynamics and fate of mercury in soils after deposition.

November 2019: NSF-funded research project to measure gaseous elemental mercury fluxes at Harvard Forest near Petersham, MA
By Eric Roy (Sophomore Environmental Science major with a focus in Atmospheric Science): I was introduced to this project through the Undergraduate Opportunities in Research and Collaborations (UROC) Immersive Scholar program at UMass Lowell, which provided me with funding to work in a research lab on campus for the summer between my freshman and sophomore years of study. As an Environmental Science major with a focus in Atmospheric Science, this project has allowed me to apply my interest in atmospheric circulation to the applied field of atmospheric chemistry. By working in the lab, I have come to realize the diverse fields of study available in atmospheric chemistry, which has encouraged me to look further into this field of research as an option for future graduate studies.
About the project:
Mercury is neurotoxic substance that has the ability to bioaccumulate in aquatic ecosystems, affecting plants, animals, and wildlife. A chemical property of Mercury (Hg) that amplifies these risks is its ability to volatilize into a gaseous form known as gaseous elemental mercury (Hg(0)). With an atmospheric residence time of up to one year, Hg(0) can be transported around the globe, making the study of the emission and deposition habits of this pollutant necessary to understand its detrimental environmental effects. Forest ecosystems are thought to be a global net sink of Hg(0), with this deposition being driven by stomatal uptake. This study aims to quantify the relative importance of both canopy and soil contributions to Hg(0) deposition and emission processes at a remote deciduous forest site at Harvard Forest, near Petersham MA. This site uses a preexisting 30 m Eddy Flux Tower to measure concentration differences both above the canopy and at the forest floor. Hg(0) concentration differences are analyzed through the calculation of time-averaged gradients and fluxes, which are then used to analyze long-term diurnal and seasonal sink and source activity.
Preliminary results indicate a net nighttime sink and a midday source of Hg(0) at both the soil and canopy levels during the photosynthetically active seasons. As a result, Hg(0) fluxes are largely decoupled from carbon dioxide, suggesting Hg(0) deposition is not driven by stomatal uptake in this forest. Also, seasonal canopy analysis indicates the forest canopy as a yearly net source of Hg(0). By obtaining a second year of data, we hope to achieve higher confidence in these preliminary trends.
By Eric Roy (Sophomore Environmental Science major with a focus in Atmospheric Science): I was introduced to this project through the Undergraduate Opportunities in Research and Collaborations (UROC) Immersive Scholar program at UMass Lowell, which provided me with funding to work in a research lab on campus for the summer between my freshman and sophomore years of study. As an Environmental Science major with a focus in Atmospheric Science, this project has allowed me to apply my interest in atmospheric circulation to the applied field of atmospheric chemistry. By working in the lab, I have come to realize the diverse fields of study available in atmospheric chemistry, which has encouraged me to look further into this field of research as an option for future graduate studies.
About the project:
Mercury is neurotoxic substance that has the ability to bioaccumulate in aquatic ecosystems, affecting plants, animals, and wildlife. A chemical property of Mercury (Hg) that amplifies these risks is its ability to volatilize into a gaseous form known as gaseous elemental mercury (Hg(0)). With an atmospheric residence time of up to one year, Hg(0) can be transported around the globe, making the study of the emission and deposition habits of this pollutant necessary to understand its detrimental environmental effects. Forest ecosystems are thought to be a global net sink of Hg(0), with this deposition being driven by stomatal uptake. This study aims to quantify the relative importance of both canopy and soil contributions to Hg(0) deposition and emission processes at a remote deciduous forest site at Harvard Forest, near Petersham MA. This site uses a preexisting 30 m Eddy Flux Tower to measure concentration differences both above the canopy and at the forest floor. Hg(0) concentration differences are analyzed through the calculation of time-averaged gradients and fluxes, which are then used to analyze long-term diurnal and seasonal sink and source activity.
Preliminary results indicate a net nighttime sink and a midday source of Hg(0) at both the soil and canopy levels during the photosynthetically active seasons. As a result, Hg(0) fluxes are largely decoupled from carbon dioxide, suggesting Hg(0) deposition is not driven by stomatal uptake in this forest. Also, seasonal canopy analysis indicates the forest canopy as a yearly net source of Hg(0). By obtaining a second year of data, we hope to achieve higher confidence in these preliminary trends.

July 2019. Research published in the Proceedings of the National Academy of Sciences of the United States of America (PNAS)
By Daniel Obrist. Obrist's research group was part of a team that reported the first direct measurements of atomic bromine in the atmosphere and confirms the important role that atomic bromine plays for depletion of ozone and mercury in the polar atmosphere.
Read the full article here: https://www.pnas.org/content/116/29/14479/tab-figures-data
Read a summary of the article here: https://cen.acs.org/environment/atmospheric-chemistry/First-direct-measurements-atomic-bromine/97/i26
By Daniel Obrist. Obrist's research group was part of a team that reported the first direct measurements of atomic bromine in the atmosphere and confirms the important role that atomic bromine plays for depletion of ozone and mercury in the polar atmosphere.
Read the full article here: https://www.pnas.org/content/116/29/14479/tab-figures-data
Read a summary of the article here: https://cen.acs.org/environment/atmospheric-chemistry/First-direct-measurements-atomic-bromine/97/i26

March 2019. Research Is Supported by $873K Grant from the National Science Foundation
By Edwin L. Aguirre (Senior Science & Technology Writer, UMass Lowell).
The National Science Foundation (NSF) has awarded a three-year, $873,000 grant to a research project led by Prof. Daniel Obrist,0 chairman of the Department of Earth, Environmental and Atmospheric Sciences, to study mercury pollution in forests.
Read the full article here: https://www.uml.edu/News/stories/2019/Obrist-NSF-grant.aspx
Funding provided by the U.S. National Science Foundation Division of Atmospheric Chemistry
By Edwin L. Aguirre (Senior Science & Technology Writer, UMass Lowell).
The National Science Foundation (NSF) has awarded a three-year, $873,000 grant to a research project led by Prof. Daniel Obrist,0 chairman of the Department of Earth, Environmental and Atmospheric Sciences, to study mercury pollution in forests.
Read the full article here: https://www.uml.edu/News/stories/2019/Obrist-NSF-grant.aspx
Funding provided by the U.S. National Science Foundation Division of Atmospheric Chemistry
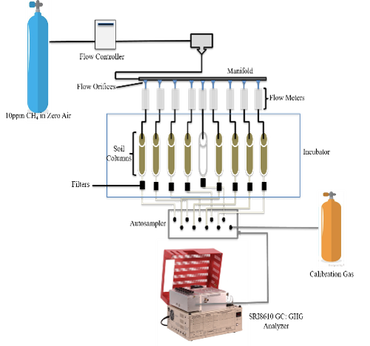
March 2019. Measuring Temperature Dependency of Methane Production in Arctic Tundra Soils
By Lea Richter (M.S. student in Graduate Program of Environmental Studies at UMass Lowell).
After carbon dioxide, atmospheric methane is the most relevant greenhouse gas affected by anthropogenic activities globally. Methane is the most abundant organic gas in the atmosphere and has a global warming potential that is ~25 times that of carbon dioxide. Climate warming in the arctic is occurring at an unprecedented rate, close to double that of lower latitudes, potentially increasing mineralization of organic carbon stored in soils and thawing permafrost and possibly enhancing methane production by methanogenic activity. On the other hand, there also is indication of enhanced methane consumption (methanotrophic activity), in particular drained upland soils that has the potential to offset methane production and even lead to soils to be net sinks of atmospheric methane .
In this study, I am assessing methane production and consumption rates, along with the production of carbon dioxide, in laboratory experiments using high-resolution flux chambers in arctic tundra soils between temperatures from -5°C to 5°C. I am using a highly controlled laboratory setup to measure high-resolution changes in methane fluxes from replicate arctic soils. Results show that carbon dioxideproduction strongly increases with temperature, with a strong step increases above freezing temperatures. Carbon dioxideproduction also shows a pronounced hysteresis effect whereby production was higher during decreasing temperatures (i.e., during freezing) compared to the same temperatures during thawing of soils. For methane, thawing soils shows some methane production (methanogenesis) upon thawing around the freezing points, possibly owed to anoxic microsites in the soils. Thereafter, soils consistently turns into net methane sinks, with higher net uptake during re-freezing of soils compared to the same temperatures upon thawing. Methanotrophy was observed at temperatures as low as -2°C after which fluxes reached near-zero.
Funding provided by U.S. Department of Energy.
By Lea Richter (M.S. student in Graduate Program of Environmental Studies at UMass Lowell).
After carbon dioxide, atmospheric methane is the most relevant greenhouse gas affected by anthropogenic activities globally. Methane is the most abundant organic gas in the atmosphere and has a global warming potential that is ~25 times that of carbon dioxide. Climate warming in the arctic is occurring at an unprecedented rate, close to double that of lower latitudes, potentially increasing mineralization of organic carbon stored in soils and thawing permafrost and possibly enhancing methane production by methanogenic activity. On the other hand, there also is indication of enhanced methane consumption (methanotrophic activity), in particular drained upland soils that has the potential to offset methane production and even lead to soils to be net sinks of atmospheric methane .
In this study, I am assessing methane production and consumption rates, along with the production of carbon dioxide, in laboratory experiments using high-resolution flux chambers in arctic tundra soils between temperatures from -5°C to 5°C. I am using a highly controlled laboratory setup to measure high-resolution changes in methane fluxes from replicate arctic soils. Results show that carbon dioxideproduction strongly increases with temperature, with a strong step increases above freezing temperatures. Carbon dioxideproduction also shows a pronounced hysteresis effect whereby production was higher during decreasing temperatures (i.e., during freezing) compared to the same temperatures during thawing of soils. For methane, thawing soils shows some methane production (methanogenesis) upon thawing around the freezing points, possibly owed to anoxic microsites in the soils. Thereafter, soils consistently turns into net methane sinks, with higher net uptake during re-freezing of soils compared to the same temperatures upon thawing. Methanotrophy was observed at temperatures as low as -2°C after which fluxes reached near-zero.
Funding provided by U.S. Department of Energy.

March 2019. Studying Mercury Dynamics in the Great Marsh of the Parker River Wildlife Estuary
By Ting Wang (Ph.D. student in the Intercampus Marine Science Program at UMass Lowell).
Among all aquatic ecosystems, salt marshes are among the most productive and play an important role at the interface between land and sea ecosystems. They provide critical ecosystems services, including as habitats for migratory birds, are important as sources and sinks for nutrients and contaminants. In my Ph.D. study, I aim to address the dynamics of mercury (Hg), an important global contaminant in the Great Marsh in the Parker River Wildlife Estuary in Massachusetts. This area, together with larger parts of New England, is a hot spot of Hg contamination (e.g. high levels of Hg observed in the blood of birds and other wildlife), although reasons for this are not fully understood. I am collecting samples of plants, soils and water to study the concentrations, mass and dynamics of Hg in this salt marsh system. One of our hypothesis is that salt marsh plants, which show a high primary productivity and can sorb Hg from atmospheric sources, serve as an important source of Hg inputs to this estuary. I am also collecting dominant plant species, including Spartina patens, Spartina alterniflora, Distichlis – spicata and Juncus – gerardi, to assess their Hg concentrations and contributions to the estuary by litterfall and plant senescence inputs. The other hypothesis that high Hg levels occur in organic-rich and old salt marsh soil, and soil as a source of Hg to tidal water (aided by tidal dynamics). I am also collecting soil depth profile in to depth of 2 meters to assess the vertical distribution of Hg and the respective mass contained in salt marsh soils. Finally, I am conducting aqueous-phase Hg sampling for both total Hg and methylated Hg along sampling transects from coastal ocean water to the Parker River estuary and the Parker River freshwater upstream to assess respective sinks and sources of Hg along this transect.
By Ting Wang (Ph.D. student in the Intercampus Marine Science Program at UMass Lowell).
Among all aquatic ecosystems, salt marshes are among the most productive and play an important role at the interface between land and sea ecosystems. They provide critical ecosystems services, including as habitats for migratory birds, are important as sources and sinks for nutrients and contaminants. In my Ph.D. study, I aim to address the dynamics of mercury (Hg), an important global contaminant in the Great Marsh in the Parker River Wildlife Estuary in Massachusetts. This area, together with larger parts of New England, is a hot spot of Hg contamination (e.g. high levels of Hg observed in the blood of birds and other wildlife), although reasons for this are not fully understood. I am collecting samples of plants, soils and water to study the concentrations, mass and dynamics of Hg in this salt marsh system. One of our hypothesis is that salt marsh plants, which show a high primary productivity and can sorb Hg from atmospheric sources, serve as an important source of Hg inputs to this estuary. I am also collecting dominant plant species, including Spartina patens, Spartina alterniflora, Distichlis – spicata and Juncus – gerardi, to assess their Hg concentrations and contributions to the estuary by litterfall and plant senescence inputs. The other hypothesis that high Hg levels occur in organic-rich and old salt marsh soil, and soil as a source of Hg to tidal water (aided by tidal dynamics). I am also collecting soil depth profile in to depth of 2 meters to assess the vertical distribution of Hg and the respective mass contained in salt marsh soils. Finally, I am conducting aqueous-phase Hg sampling for both total Hg and methylated Hg along sampling transects from coastal ocean water to the Parker River estuary and the Parker River freshwater upstream to assess respective sinks and sources of Hg along this transect.

February 2018. Study on Atmospheric Mercury Uptake Published in 'Nature Geoscience'
By Daniel Obrist. An new article entitled "A vegetation control on seasonal variations in global atmospheric mercury concentrations" was published in 'Nature Geoscience'. The study was led by colleagues from the University of Basel, Switzerland (Dr. Martin Jiskra) and CNRS in France (Dr. Jeroen Sonke) in collaboration with our group. We now understand that the dominant source of mercury in many terrestrial ecosystems is from atmospheric elemental mercury deposition taken up by vegetation and transferred to soils when plants die off or shed leaves (part of this is termed “litterfall”. Our study estimated that this uptake of atmospheric mercury and subsequent deposition is the dominant pathway of mercury inputs to terrestrial ecosystems, globally accounting for over 1,000 Mg(0) – or 20% to 25% of the total atmospheric Hg pool – each year. We further show that the strong global vegetation mercury sink (or “pump”) induces pronounced seasonal variability and drives diurnal variation of atmospheric mercury concentrations. For example, in summertime, atmospheric mercury minima at remote sites have previously been explained by oxidation chemistry and seasonality of emissions, but can be better explained by terrestrial ecosystems sequestration of mercury via plant uptake.
Full reference: Martin M, Sonke JE, Obrist D, Bieser J, Ebinghaus R, Myhre CL, Pfaffhuber, KA, Wängberg I, Kyllönen K, Worthy D, Martin LG, Labuschagne C, Mkololo T, Ramonet M, Magand O, Dommergue A. A vegetation control on seasonal variations in global atmospheric mercury. Nature Geoscience, 11, 244-250, 2018.
Funding was in part provided by NSF.
By Daniel Obrist. An new article entitled "A vegetation control on seasonal variations in global atmospheric mercury concentrations" was published in 'Nature Geoscience'. The study was led by colleagues from the University of Basel, Switzerland (Dr. Martin Jiskra) and CNRS in France (Dr. Jeroen Sonke) in collaboration with our group. We now understand that the dominant source of mercury in many terrestrial ecosystems is from atmospheric elemental mercury deposition taken up by vegetation and transferred to soils when plants die off or shed leaves (part of this is termed “litterfall”. Our study estimated that this uptake of atmospheric mercury and subsequent deposition is the dominant pathway of mercury inputs to terrestrial ecosystems, globally accounting for over 1,000 Mg(0) – or 20% to 25% of the total atmospheric Hg pool – each year. We further show that the strong global vegetation mercury sink (or “pump”) induces pronounced seasonal variability and drives diurnal variation of atmospheric mercury concentrations. For example, in summertime, atmospheric mercury minima at remote sites have previously been explained by oxidation chemistry and seasonality of emissions, but can be better explained by terrestrial ecosystems sequestration of mercury via plant uptake.
Full reference: Martin M, Sonke JE, Obrist D, Bieser J, Ebinghaus R, Myhre CL, Pfaffhuber, KA, Wängberg I, Kyllönen K, Worthy D, Martin LG, Labuschagne C, Mkololo T, Ramonet M, Magand O, Dommergue A. A vegetation control on seasonal variations in global atmospheric mercury. Nature Geoscience, 11, 244-250, 2018.
Funding was in part provided by NSF.

July 2017. Obrist's Research Published in 'Nature'
Press Release, Written By Edwin Aguirre. Large quantities of mercury are ending up in the Arctic, threatening the environment and the health and well-being of northern wildlife and people, according to new research led by Prof. Daniel Obrist, chairman of the Department of Environmental, Earth and Atmospheric Sciences. In a study published this month in the journal Nature, Obrist’s team found that the absorption of mercury from the atmosphere by the tundra is shown to drive high loads of mercury in Arctic tundra soils. Tundras are vast, treeless regions where the soils are permanently frozen, or “permafrost.” Mercury runoff from tundra soil then supplies 65 to 85 tons of the toxic heavy metal to Arctic lakes, rivers and the Arctic Ocean each year. “This mercury from the tundra soil explains half to two-thirds of the total mercury input to the Arctic Ocean,” says Obrist.
Obrist recently led an international group — including researchers from UMass Lowell, the Desert Research Institute in Reno, Nev., Paris-Sorbonne University and the University of Toulouse in France, the University of Colorado and the Gas Technology Institute in Des Plaines, Ill. — to conduct a long-term study of the origin of mercury pollution in the Arctic tundra. The investigation was supported by the U.S. National Science Foundation. “This is the most comprehensive study ever done on mercury deposition,” notes Obrist. Mercury is one of the most harmful pollutants threatening fish, birds and mammals worldwide. Hundreds of tons of it are released into the atmosphere each year by power plants through the burning of coal, as well as through mining activities and other industrial processes, and are distributed throughout the world. Airborne mercury eventually falls back to the ground, accumulating in soils, rivers, lakes and oceans. A first global treaty, the Minamata Convention that aims to protect human health and the environment from the adverse effects of mercury, was adopted in 2013. It has been signed by 128 member countries — including the U.S. — and will take effect in August of this year. “Mercury is a neurotoxic pollutant that has led to high exposure levels in northern wildlife, such as beluga whales, polar bears, seals, fish, eagles and other seabirds,” says Obrist. “It also affects human populations, particularly the Inuit, which rely on traditional hunting and fishing.” Exposure to high levels of mercury over long periods can lead to neurological and cardiovascular problems. Infants and young children can suffer from developmental disorders, reduced memory performance and increased risk of attention problems.
Mercury exists in several forms, including methylmercury and other organic compounds, elemental (metallic) mercury and inorganic mercury compounds. “We determined the dominant source of mercury in the Arctic tundra to be the gaseous form of elemental mercury that is being transported in the atmosphere,” says Obrist. “During a span of two years, we developed a complete accounting of all atmospheric mercury inputs and an inventory of soil mercury deposits at our observation site, which is in an inland tundra in northern Alaska north of the Brooks Range. We used a unique combination of different techniques that allowed us to make precise measurements for two full years, including through the Arctic winter.” The scientists found that 70 percent of atmospheric mercury deposition is driven by the gaseous elemental form, whose deposition is normally not measured and travels the farthest distances in the atmosphere so it easily reaches the Arctic. In contrast, deposition via rain and snow, which generally is the focus in most studies, is very small over the northern Arctic tundra — approximately 2 percent of the total mercury deposition. How does gaseous elemental mercury end up in the tundra soil? “It’s a process similar to the uptake of carbon dioxide by plants. In this case, the mercury gas is absorbed by tundra plants and then transferred to the soil when the plants shed leaves or die off,” explains Obrist. As a result, the Arctic tundra becomes a significant deposit for mercury emitted from industrialized, more southern regions of the world. “We calculate that large amounts of mercury are trapped in tundra soils, possibly up to a third to one-half of the world’s total soil mercury deposits. These deposits may be critically threatened by an unprecedented Arctic climate warming that could destabilize Arctic soils and possibly lead to more mercury being released into streams, rivers, lakes and the Arctic Ocean. This would further harm the welfare of Arctic wildlife and people,” adds Obrist.
Full reference: Obrist D, Agnan Y, Jiskra M, Hedge C, Colegrove D, Hueber J, Moore C, Sonke J, Helmig D. Tundra uptake of atmospheric elemental mercury drives arctic mercury pollution. Nature, 547, 201-204, 2017.
Funding provided by NSF Polar Program and the Department of Energy.
Press Release, Written By Edwin Aguirre. Large quantities of mercury are ending up in the Arctic, threatening the environment and the health and well-being of northern wildlife and people, according to new research led by Prof. Daniel Obrist, chairman of the Department of Environmental, Earth and Atmospheric Sciences. In a study published this month in the journal Nature, Obrist’s team found that the absorption of mercury from the atmosphere by the tundra is shown to drive high loads of mercury in Arctic tundra soils. Tundras are vast, treeless regions where the soils are permanently frozen, or “permafrost.” Mercury runoff from tundra soil then supplies 65 to 85 tons of the toxic heavy metal to Arctic lakes, rivers and the Arctic Ocean each year. “This mercury from the tundra soil explains half to two-thirds of the total mercury input to the Arctic Ocean,” says Obrist.
Obrist recently led an international group — including researchers from UMass Lowell, the Desert Research Institute in Reno, Nev., Paris-Sorbonne University and the University of Toulouse in France, the University of Colorado and the Gas Technology Institute in Des Plaines, Ill. — to conduct a long-term study of the origin of mercury pollution in the Arctic tundra. The investigation was supported by the U.S. National Science Foundation. “This is the most comprehensive study ever done on mercury deposition,” notes Obrist. Mercury is one of the most harmful pollutants threatening fish, birds and mammals worldwide. Hundreds of tons of it are released into the atmosphere each year by power plants through the burning of coal, as well as through mining activities and other industrial processes, and are distributed throughout the world. Airborne mercury eventually falls back to the ground, accumulating in soils, rivers, lakes and oceans. A first global treaty, the Minamata Convention that aims to protect human health and the environment from the adverse effects of mercury, was adopted in 2013. It has been signed by 128 member countries — including the U.S. — and will take effect in August of this year. “Mercury is a neurotoxic pollutant that has led to high exposure levels in northern wildlife, such as beluga whales, polar bears, seals, fish, eagles and other seabirds,” says Obrist. “It also affects human populations, particularly the Inuit, which rely on traditional hunting and fishing.” Exposure to high levels of mercury over long periods can lead to neurological and cardiovascular problems. Infants and young children can suffer from developmental disorders, reduced memory performance and increased risk of attention problems.
Mercury exists in several forms, including methylmercury and other organic compounds, elemental (metallic) mercury and inorganic mercury compounds. “We determined the dominant source of mercury in the Arctic tundra to be the gaseous form of elemental mercury that is being transported in the atmosphere,” says Obrist. “During a span of two years, we developed a complete accounting of all atmospheric mercury inputs and an inventory of soil mercury deposits at our observation site, which is in an inland tundra in northern Alaska north of the Brooks Range. We used a unique combination of different techniques that allowed us to make precise measurements for two full years, including through the Arctic winter.” The scientists found that 70 percent of atmospheric mercury deposition is driven by the gaseous elemental form, whose deposition is normally not measured and travels the farthest distances in the atmosphere so it easily reaches the Arctic. In contrast, deposition via rain and snow, which generally is the focus in most studies, is very small over the northern Arctic tundra — approximately 2 percent of the total mercury deposition. How does gaseous elemental mercury end up in the tundra soil? “It’s a process similar to the uptake of carbon dioxide by plants. In this case, the mercury gas is absorbed by tundra plants and then transferred to the soil when the plants shed leaves or die off,” explains Obrist. As a result, the Arctic tundra becomes a significant deposit for mercury emitted from industrialized, more southern regions of the world. “We calculate that large amounts of mercury are trapped in tundra soils, possibly up to a third to one-half of the world’s total soil mercury deposits. These deposits may be critically threatened by an unprecedented Arctic climate warming that could destabilize Arctic soils and possibly lead to more mercury being released into streams, rivers, lakes and the Arctic Ocean. This would further harm the welfare of Arctic wildlife and people,” adds Obrist.
Full reference: Obrist D, Agnan Y, Jiskra M, Hedge C, Colegrove D, Hueber J, Moore C, Sonke J, Helmig D. Tundra uptake of atmospheric elemental mercury drives arctic mercury pollution. Nature, 547, 201-204, 2017.
Funding provided by NSF Polar Program and the Department of Energy.

August 2015. Mercury isotope measurements at Toolik Field Station
By Martin Jiskra. During the visit to Toolik Field Station in August, Daniel and Martin installed a new setup to collect air samples for mercury isotope measurements. Martin works as a Postdoc at Geosciences Environment Toulouse in France (CNRS) on a project called: "Understanding the fate of Arctic atmospheric mercury (Hg) deposition – A Hg stable isotope investigation of redox processes and Hg re-emissions" in collaboration with Daniel’s group. The sampling campaign will run through the winter for one year. The goal of the project is to measure the variation of stable mercury isotopes at Toolik Field station in the Arctic tundra in order to gain additional insights into the processes governing the mercury exchange between the atmosphere and snow and soils.
Funding provided by EU Horizon 2020 (Marie Curie fellowship to Martin Jiskra)
By Martin Jiskra. During the visit to Toolik Field Station in August, Daniel and Martin installed a new setup to collect air samples for mercury isotope measurements. Martin works as a Postdoc at Geosciences Environment Toulouse in France (CNRS) on a project called: "Understanding the fate of Arctic atmospheric mercury (Hg) deposition – A Hg stable isotope investigation of redox processes and Hg re-emissions" in collaboration with Daniel’s group. The sampling campaign will run through the winter for one year. The goal of the project is to measure the variation of stable mercury isotopes at Toolik Field station in the Arctic tundra in order to gain additional insights into the processes governing the mercury exchange between the atmosphere and snow and soils.
Funding provided by EU Horizon 2020 (Marie Curie fellowship to Martin Jiskra)
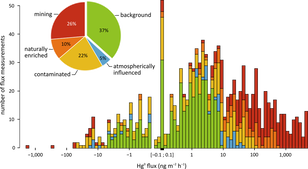
August 2015. New constraints on terrestrial surface-atmosphere fluxes of gaseous elemental mercury using a global database
By Yannick Agnan. Gaseous elemental mercury (Hg0) is a semi volatile chemical easily exchanged between terrestrial surfaces and the atmosphere. In spite of nearly 40 years of research, major uncertainties still exist concerning factors controlling this exchange and the influence of methodology used (dynamic flux chambers and micrometeorological methods). 132 published papers were compiled in a global database, including ca. 200,000 estimated Hg0 flux measurements. The authors (Y. Agnan, T. Le Dantec, C. Moore, G. Edwards, and D. Obrist) showed that measurements were unevenly distributed. Fluxes measured in Hg enriched sites were driven by substrate Hg concentrations, as observed by a log relationship which is absent in background areas. Flux over litter- and snow-covered soils were lower than measurements over bare soils. The annual worldwide estimates based on land covers were 523 Mg a−1 (with a large 25% uncertainties from −592 to 1,557 Mg a−1), including 129 Mg a−1 for background landscapes, 261 Mg a−1 for atmospherically-influenced sites (low substrate Hg concentration, but atmospheric Hg0 concentration > 3 ng m−3), and 133 Mg a−1 for contaminated, naturally-enriched, and mining areas.
The full reference of the article is: Agnan Y., Le Dantec T., Moore CW, Edwards GC, Obrist D. New constraints on terrestrial surface-atmosphere fluxes of gaseous elemental mercury using a global database. Environmental Science and Technology, 50, 507-524, 2015.
Funding provided by NFS (Coupled Natural and Human Systems)
By Yannick Agnan. Gaseous elemental mercury (Hg0) is a semi volatile chemical easily exchanged between terrestrial surfaces and the atmosphere. In spite of nearly 40 years of research, major uncertainties still exist concerning factors controlling this exchange and the influence of methodology used (dynamic flux chambers and micrometeorological methods). 132 published papers were compiled in a global database, including ca. 200,000 estimated Hg0 flux measurements. The authors (Y. Agnan, T. Le Dantec, C. Moore, G. Edwards, and D. Obrist) showed that measurements were unevenly distributed. Fluxes measured in Hg enriched sites were driven by substrate Hg concentrations, as observed by a log relationship which is absent in background areas. Flux over litter- and snow-covered soils were lower than measurements over bare soils. The annual worldwide estimates based on land covers were 523 Mg a−1 (with a large 25% uncertainties from −592 to 1,557 Mg a−1), including 129 Mg a−1 for background landscapes, 261 Mg a−1 for atmospherically-influenced sites (low substrate Hg concentration, but atmospheric Hg0 concentration > 3 ng m−3), and 133 Mg a−1 for contaminated, naturally-enriched, and mining areas.
The full reference of the article is: Agnan Y., Le Dantec T., Moore CW, Edwards GC, Obrist D. New constraints on terrestrial surface-atmosphere fluxes of gaseous elemental mercury using a global database. Environmental Science and Technology, 50, 507-524, 2015.
Funding provided by NFS (Coupled Natural and Human Systems)
 Soil pit (top) and portion of a permafrost core (bottom) sampled along the Dalton Highway transect.
Soil pit (top) and portion of a permafrost core (bottom) sampled along the Dalton Highway transect.
August 2015. Dalton Highway transect sampling in northern Alaska to provide new insights into spatial distribution of mercury in vegetation and soils of tundra ecosystems
By Christine Hedge. Christine traveled to northern Alaska for her first field visit with Daniel to perform sampling for her PhD research. In addition to sampling vegetation, soil, and runoff at the Toolik Field Station, sampling was done along a northern transect extending from Toolik Field Station to the Arctic Ocean along the Dalton Highway. Eight sites in total were sampled along the transect in areas where previous soils data on carbon, nitrogen, and other components had been measured to leverage this investigation upon. Analysis of Hg in the terrestrial landscape of these tundra sites will allow Christine to investigate the spatial distribution of mercury from an inland to coastal region in an area where almost no data on Hg within soils exists. Additional investigations from this transect sampling will include understanding how atmospheric deposition of Hg may change along this gradient and how other factors such as soil age, soil depth, carbon content, soil chemistry, and plant cover may influence storage and cycling of Hg within tundra ecosystems.
Soil pit (top) and portion of a permafrost core (bottom) sampled along the Dalton Highway transect.
Funding provided by NFS (Polar Programs) and Alaska National Park Service
By Christine Hedge. Christine traveled to northern Alaska for her first field visit with Daniel to perform sampling for her PhD research. In addition to sampling vegetation, soil, and runoff at the Toolik Field Station, sampling was done along a northern transect extending from Toolik Field Station to the Arctic Ocean along the Dalton Highway. Eight sites in total were sampled along the transect in areas where previous soils data on carbon, nitrogen, and other components had been measured to leverage this investigation upon. Analysis of Hg in the terrestrial landscape of these tundra sites will allow Christine to investigate the spatial distribution of mercury from an inland to coastal region in an area where almost no data on Hg within soils exists. Additional investigations from this transect sampling will include understanding how atmospheric deposition of Hg may change along this gradient and how other factors such as soil age, soil depth, carbon content, soil chemistry, and plant cover may influence storage and cycling of Hg within tundra ecosystems.
Soil pit (top) and portion of a permafrost core (bottom) sampled along the Dalton Highway transect.
Funding provided by NFS (Polar Programs) and Alaska National Park Service

August 2015. Arctic Hg cycling project at Toolik Field station in northern Alaska enters second year of continuous field measurement
By Daniel Obrist. Christine and Daniel recently visited Toolik Field station to re-set and calibrate systems and instruments to begin a second year of field measurements on the north-slope of the Brooks Range in northern Alaska. As part of a project entitled “Collaborative Research: Soil−Snow−Atmosphere Exchange of Mercury in the Interior Arctic Tundra” (collaboration with D. Helmig’s group at the University of Colorado, Boulder), we have been measuring trace gas exchanges of Hg and other trace gases between soils, snow, plants, and the atmosphere for over a year. In addition, we are characterizing other deposition fluxes, soil and snow chemistry, and soil solution and stream characterization. We are excited to enter the second winter of our observations, and are in the process of analysis of a first full year of unique observations in the arctic tundra.
Funding provided by NFS (Polar Programs)
By Daniel Obrist. Christine and Daniel recently visited Toolik Field station to re-set and calibrate systems and instruments to begin a second year of field measurements on the north-slope of the Brooks Range in northern Alaska. As part of a project entitled “Collaborative Research: Soil−Snow−Atmosphere Exchange of Mercury in the Interior Arctic Tundra” (collaboration with D. Helmig’s group at the University of Colorado, Boulder), we have been measuring trace gas exchanges of Hg and other trace gases between soils, snow, plants, and the atmosphere for over a year. In addition, we are characterizing other deposition fluxes, soil and snow chemistry, and soil solution and stream characterization. We are excited to enter the second winter of our observations, and are in the process of analysis of a first full year of unique observations in the arctic tundra.
Funding provided by NFS (Polar Programs)
July 2015. Participation and publication with the Western North American Mercury Synthesis Working Group
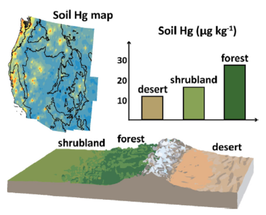
By Daniel Obrist. Daniel was involved in a synthesis effort by the “Western North American Mercury Synthesis Working Group” to assess mercury impacts across the west. As part of this effort, he attended two workshops at the John Wesley Powell Center for Analysis and Synthesis in Fort Collins, CO, and took the lead on a focus paper describing terrestrial mercury distribution patterns across nine states of the contiguous Western United States. His manuscript entitled “Terrestrial mercury in the western United States: spatial distribution defined by land cover and plant productivity” and authored by several colleagues (Daniel Obrist, Chris Pearson, Jackson Webster, Tyler Kane; Che-Jen Lin; George R. Aiken, and Charles N. Alpers) was recently submitted to Science of the Total Environment. A key result of the analysis is that vegetation patterns have strong effects on spatial distribution patterns of soil mercury across the Western U.S., likely driven by plant productivity. These effects result in 2.5 times higher soil Hg concentrations in forested areas compared to barren locations.
The full reference of the article is: Obrist D, Pearson C, Webster J, Kane T, Lin C-L, Aiken GR, Alpers CN Terrestrial mercury in the western United States: spatial distribution defined by land cover and plant productivity. The Science of the Total Environment, 568, 522-535, 2016.
Funding provided by the DRI (Division of Atmospheric Sciences) and the USGS
By Daniel Obrist. Daniel was involved in a synthesis effort by the “Western North American Mercury Synthesis Working Group” to assess mercury impacts across the west. As part of this effort, he attended two workshops at the John Wesley Powell Center for Analysis and Synthesis in Fort Collins, CO, and took the lead on a focus paper describing terrestrial mercury distribution patterns across nine states of the contiguous Western United States. His manuscript entitled “Terrestrial mercury in the western United States: spatial distribution defined by land cover and plant productivity” and authored by several colleagues (Daniel Obrist, Chris Pearson, Jackson Webster, Tyler Kane; Che-Jen Lin; George R. Aiken, and Charles N. Alpers) was recently submitted to Science of the Total Environment. A key result of the analysis is that vegetation patterns have strong effects on spatial distribution patterns of soil mercury across the Western U.S., likely driven by plant productivity. These effects result in 2.5 times higher soil Hg concentrations in forested areas compared to barren locations.
The full reference of the article is: Obrist D, Pearson C, Webster J, Kane T, Lin C-L, Aiken GR, Alpers CN Terrestrial mercury in the western United States: spatial distribution defined by land cover and plant productivity. The Science of the Total Environment, 568, 522-535, 2016.
Funding provided by the DRI (Division of Atmospheric Sciences) and the USGS

June 2015. Participation in the 12th annual International Conference of Mercury as a Global Pollutant in Jeju, South Korea
By Christine Hedge. Christine attended the 12th International Conference of Mercury as a Global Polluant in Jeju, South Korea where she gave an oral presentation entitled “Soil and Plant Mercury Concentration and pools in northern Alaska. Her presentation included initial results of analysis on soil and vegetation from the Toolik Field Station (TFS) in northern Alaska, the main study site of her project. Highlights from the initial results showed surprisingly high plant Hg deposition rates of 17.3 µg m−2 yr−1 which is comparable to rates found in temperate regions. Within the soils, organic Hg concentrations at TFS are similar to what is observed in temperate regions however the mineral horizons show concentrations of 2−3 times what is observed in the lower 48 states. Over 90% of the mercury is stored in the mineral layers of soil at Toolik which is a very high pool of mercury that we may have to be concerned about with thawing of permafrost soils.
By Christine Hedge. Christine attended the 12th International Conference of Mercury as a Global Polluant in Jeju, South Korea where she gave an oral presentation entitled “Soil and Plant Mercury Concentration and pools in northern Alaska. Her presentation included initial results of analysis on soil and vegetation from the Toolik Field Station (TFS) in northern Alaska, the main study site of her project. Highlights from the initial results showed surprisingly high plant Hg deposition rates of 17.3 µg m−2 yr−1 which is comparable to rates found in temperate regions. Within the soils, organic Hg concentrations at TFS are similar to what is observed in temperate regions however the mineral horizons show concentrations of 2−3 times what is observed in the lower 48 states. Over 90% of the mercury is stored in the mineral layers of soil at Toolik which is a very high pool of mercury that we may have to be concerned about with thawing of permafrost soils.

June 2015. New paper on snowpack nutrient and pollutant loads in the Lake Tahoe basin
By Daniel Obrist. Chris Pearson (former Graduate Student in Graduate Program of Hydrologic Sciences) is the first author of a new article published in Biogeosciences entitled “Nutrient and mercury deposition and storage in an alpine snowpack of the Sierra Nevada, USA”. Chris and his co-authors (Rina Schumer, Ben Trustman, Karl Rittger, Dale Johnson, and Daniel Obrist) chemically characterized snowpack in the Lake Tahoe basin over the duration of two winters. Their results show a key importance of organic nitrogen composing on average almost 50% of total nitrogen observed in snowpack. Detailed spatial and temporal snowpack sampling revealed that nitrate was supplied relatively evenly across the basin via w et deposition, and that ammonia/ nitrogen was driven by dry deposition with was particularly enhanced in late winter and spring and along the western side of the basin. Snowpack phosphorus was strongly enhanced near urban areas in the Lake Tahoe basin. Mercury showed low but detectable snowpack levels, and the fact that snowpack concentrations were significantly lower lower compared to measured wet deposition revealed an important role of photochemical re-emissions after snow-related deposition.
The full reference of the article is: Pearson C, Schumer R, Rittger K, Johnson DW, Obrist D. Nutrient and mercury deposition and storage in an alpine snowpack of the Sierra Nevada, USA. Biogeosciences 12, 3665-3680, 2015.
Funding provided by the USGS (State Water Research Program)
By Daniel Obrist. Chris Pearson (former Graduate Student in Graduate Program of Hydrologic Sciences) is the first author of a new article published in Biogeosciences entitled “Nutrient and mercury deposition and storage in an alpine snowpack of the Sierra Nevada, USA”. Chris and his co-authors (Rina Schumer, Ben Trustman, Karl Rittger, Dale Johnson, and Daniel Obrist) chemically characterized snowpack in the Lake Tahoe basin over the duration of two winters. Their results show a key importance of organic nitrogen composing on average almost 50% of total nitrogen observed in snowpack. Detailed spatial and temporal snowpack sampling revealed that nitrate was supplied relatively evenly across the basin via w et deposition, and that ammonia/ nitrogen was driven by dry deposition with was particularly enhanced in late winter and spring and along the western side of the basin. Snowpack phosphorus was strongly enhanced near urban areas in the Lake Tahoe basin. Mercury showed low but detectable snowpack levels, and the fact that snowpack concentrations were significantly lower lower compared to measured wet deposition revealed an important role of photochemical re-emissions after snow-related deposition.
The full reference of the article is: Pearson C, Schumer R, Rittger K, Johnson DW, Obrist D. Nutrient and mercury deposition and storage in an alpine snowpack of the Sierra Nevada, USA. Biogeosciences 12, 3665-3680, 2015.
Funding provided by the USGS (State Water Research Program)
March 2015. Paper on polycyclic aromatic hydrocarbons in forest soils accepted in Chemosphere
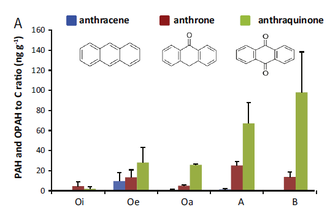
By Daniel Obrist. A new article was published in Chemosphere. entitled “Accumulation of polycyclic aromatic hydrocarbons (PAHs) and oxygenated PAHs (OPAHs) in organic and mineral soil horizons from four U.S. remote forests”. In this article, authored by Daniel Obrist, Barbara Zielinksa (both DRI), and Judith Perlinger (Michigan Technological University), we describe the distribution of 23 PAH and 9 OPAH compounds across four remote forests of the United States. We found that forest soils accumulate significant levels of PAHs in litter and soil horizons, and that the accumulation was related to the water solubility of compounds. Highly toxic OPAHs, which are also emitted during combustion processes and in addition form in-situ in soils, show higher accumulation compared to their parent-PAHs, particularly in decomposed litter and in soil layers. These patterns suggest that OPAHs have a high persistency in litter and soils and a strong sorption behavior to organic matter.
The full reference of the article is: Obrist D, Zielinska B, Perlinger J. Accumulation of polycyclic aromatic hydrocarbons (PAHs) and oxygenated PAHs (OPAHs) in organic and mineral soil horizons from four U.S. remote forests. Chemosphere 134, 98-105, 2015.
Funding provided by the DRI (Division of Atmospheric Sciences) and EPA STAR
By Daniel Obrist. A new article was published in Chemosphere. entitled “Accumulation of polycyclic aromatic hydrocarbons (PAHs) and oxygenated PAHs (OPAHs) in organic and mineral soil horizons from four U.S. remote forests”. In this article, authored by Daniel Obrist, Barbara Zielinksa (both DRI), and Judith Perlinger (Michigan Technological University), we describe the distribution of 23 PAH and 9 OPAH compounds across four remote forests of the United States. We found that forest soils accumulate significant levels of PAHs in litter and soil horizons, and that the accumulation was related to the water solubility of compounds. Highly toxic OPAHs, which are also emitted during combustion processes and in addition form in-situ in soils, show higher accumulation compared to their parent-PAHs, particularly in decomposed litter and in soil layers. These patterns suggest that OPAHs have a high persistency in litter and soils and a strong sorption behavior to organic matter.
The full reference of the article is: Obrist D, Zielinska B, Perlinger J. Accumulation of polycyclic aromatic hydrocarbons (PAHs) and oxygenated PAHs (OPAHs) in organic and mineral soil horizons from four U.S. remote forests. Chemosphere 134, 98-105, 2015.
Funding provided by the DRI (Division of Atmospheric Sciences) and EPA STAR

March 2015. Funding provided by the Alaska National Park Service to extend research on the terrestrial cycling of mercury to national parks in northern Alaska
By Christine Hedge. Christine was awarded a scholarship from the Alaska National Park Service and Murie Science and Learning Center to extend her research on mercury pollutant cycling in tundra ecosystems to include areas in and near parks in northern Alaska. Sampling locations include Denali National Park, the Noatak National Preserve, and the Arctic National Wildlife Refuge. The first research objective is to understand how Hg is distributed spatially across different tundra systems of Alaska, in particular in soils and plants, to determine what areas are affected most by atmospheric deposition. Second, is to quantify the pools of Hg stored in tundra ecosystems to understand if tundra ecosystems serve as important repositories, storing current and past atmospheric Hg pollution that has been transported to the Arctic. Finally, she will study the link between soil Hg concentrations and Hg in aqueous phase (soil solution and adjacent streams) to assess how runoff is linked to upland soil concentrations and understand what controls the transport and mobilization of this pollutant across different tundra regions.
Funding provided by NFS (Polar Programs) and Alaska National Park Service
By Christine Hedge. Christine was awarded a scholarship from the Alaska National Park Service and Murie Science and Learning Center to extend her research on mercury pollutant cycling in tundra ecosystems to include areas in and near parks in northern Alaska. Sampling locations include Denali National Park, the Noatak National Preserve, and the Arctic National Wildlife Refuge. The first research objective is to understand how Hg is distributed spatially across different tundra systems of Alaska, in particular in soils and plants, to determine what areas are affected most by atmospheric deposition. Second, is to quantify the pools of Hg stored in tundra ecosystems to understand if tundra ecosystems serve as important repositories, storing current and past atmospheric Hg pollution that has been transported to the Arctic. Finally, she will study the link between soil Hg concentrations and Hg in aqueous phase (soil solution and adjacent streams) to assess how runoff is linked to upland soil concentrations and understand what controls the transport and mobilization of this pollutant across different tundra regions.
Funding provided by NFS (Polar Programs) and Alaska National Park Service
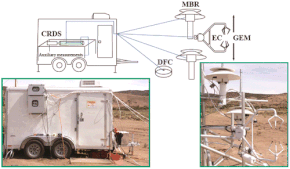
February 2015. First-ever eddy covariance measurements of gaseous elemental mercury published in Environmental Science & Technology
By Chris Moore. Ashley Pierce (former graduate student) was first author and led the publication in Environmental Science & Technology of results from a field campaign that we conducted in Palomino Valley, NV using our newly developed cavity ring-down spectrometer system. Publication of this data was extremely challenging and involved a whole team of international scientists, including Chris Moore (DRI), Georg Wohlfahrt (University of Innsbruck, Austria), Lukas Hortnagl (ETH Zurich, Switzerland), Natascha Kljun (Swansea University, U.K.) and Daniel Obrist (DRI). The results are extremely important as until this system measurement of gaseous elemental mercury fluxes via the highly effective eddy covariance technique was out of reach of current instrumentation. However, these measurements were made over contaminated sources and more work is needed to get the sensitivity needed for background soils/surfaces.
Funding provided by NFS (Major Instrumentation)
By Chris Moore. Ashley Pierce (former graduate student) was first author and led the publication in Environmental Science & Technology of results from a field campaign that we conducted in Palomino Valley, NV using our newly developed cavity ring-down spectrometer system. Publication of this data was extremely challenging and involved a whole team of international scientists, including Chris Moore (DRI), Georg Wohlfahrt (University of Innsbruck, Austria), Lukas Hortnagl (ETH Zurich, Switzerland), Natascha Kljun (Swansea University, U.K.) and Daniel Obrist (DRI). The results are extremely important as until this system measurement of gaseous elemental mercury fluxes via the highly effective eddy covariance technique was out of reach of current instrumentation. However, these measurements were made over contaminated sources and more work is needed to get the sensitivity needed for background soils/surfaces.
Funding provided by NFS (Major Instrumentation)

July 2013. Atmospheric simulation chamber for studying mercury chemistry
By Steven Darby/Ashley Pierce. Mercury is a pollutant that can be transported easily through the atmosphere. Typically the concentration in the atmosphere is low – the problems start when mercury is deposited to the surface where it may bioaccumulate through the food chain. The mechanism of deposition is not well known, but some studies have indicated that reactions involving bromine are key. These studies are often carried out at room temperature, with very large concentrations that are not found in the environment. In this project we tested various mercury reactions directly at low concentrations and low temperatures that are more representative of the conditions in the environment. To do this we constructed a 300 L FEP chamber, temperature controlled between −10 to +30°C. A Cavity Ring-Down Spectroscopy (CRDS) system quantifies gaseous elemental mercury (GEM) in situ with a time resolution of 25 Hz. Two more optical cavities use Incoherent Broadband Cavity-Enhanced Absorption Spectroscopy (IBBCEAS) to measure Br2, NO2, and BrO. Measurements also included the addition of NO2, O3 and sea salt aerosols into the chamber with GEM and Br2. The goal of this project was to better quantify the reaction kinetics for these compounds.
Funding provided by Science Foundation Ireland and NSF (Major Instrumentation)
By Steven Darby/Ashley Pierce. Mercury is a pollutant that can be transported easily through the atmosphere. Typically the concentration in the atmosphere is low – the problems start when mercury is deposited to the surface where it may bioaccumulate through the food chain. The mechanism of deposition is not well known, but some studies have indicated that reactions involving bromine are key. These studies are often carried out at room temperature, with very large concentrations that are not found in the environment. In this project we tested various mercury reactions directly at low concentrations and low temperatures that are more representative of the conditions in the environment. To do this we constructed a 300 L FEP chamber, temperature controlled between −10 to +30°C. A Cavity Ring-Down Spectroscopy (CRDS) system quantifies gaseous elemental mercury (GEM) in situ with a time resolution of 25 Hz. Two more optical cavities use Incoherent Broadband Cavity-Enhanced Absorption Spectroscopy (IBBCEAS) to measure Br2, NO2, and BrO. Measurements also included the addition of NO2, O3 and sea salt aerosols into the chamber with GEM and Br2. The goal of this project was to better quantify the reaction kinetics for these compounds.
Funding provided by Science Foundation Ireland and NSF (Major Instrumentation)

April 2013. Development of a cavity ring-down spectroscopy sensor for measuring atmospheric gaseous elemental mercury concentrations
By Ashley Pierce. Due to our lack of understanding of parts of the global Hg cycle it is important to improve available measurement techniques. Current instruments for measuring Hg and specifically gaseous elemental mercury (GEM) take several minutes to analyze and sever liters of air per sample. GEM has a longer atmospheric residence time than other forms of Hg and this coupled with its semi-volatile nature means GEM can travel long distances where it can then be deposited to remote ecosystems and eventually transformed into bioavailable toxic forms of Hg (i.e. methylmercury). Deposition is the main source of Hg to remote ecosystems. Our lab has developed a cavity ring-down spectroscopy (CRDS) sensor for measuring atmospheric GEM concentrations. Using a laser system, that outputs the wavelength that GEM absorbs (253.65 nm), we coupled the laser beam into a measurement cavity with a mirror at each end. The laser pulse bounces between the two mirrors effectively creating a long path length and high sensitivity while keeping the sample size small. Differential measurements allow for continuous correction of system baseline extinction losses. The CRDS sensor is used to measure GEM in ambient air and laboratory studies at 25 Hz, as well as to measure GEM fluxes using the eddy covariance (EC) flux measurement technique which is the first application of EC for GEM. With these measurements we aim to improve the quantification of dry deposition of GEM which is confounded by the bi-directional movement of GEM between surfaces and the atmosphere, to better measure fast concentration fluctuations from sources and sinks and during chemical transformations. Results of this study are published in Atmospheric Measurement Techniques.
Funding provided by NSF (Major Instrumentation)
By Ashley Pierce. Due to our lack of understanding of parts of the global Hg cycle it is important to improve available measurement techniques. Current instruments for measuring Hg and specifically gaseous elemental mercury (GEM) take several minutes to analyze and sever liters of air per sample. GEM has a longer atmospheric residence time than other forms of Hg and this coupled with its semi-volatile nature means GEM can travel long distances where it can then be deposited to remote ecosystems and eventually transformed into bioavailable toxic forms of Hg (i.e. methylmercury). Deposition is the main source of Hg to remote ecosystems. Our lab has developed a cavity ring-down spectroscopy (CRDS) sensor for measuring atmospheric GEM concentrations. Using a laser system, that outputs the wavelength that GEM absorbs (253.65 nm), we coupled the laser beam into a measurement cavity with a mirror at each end. The laser pulse bounces between the two mirrors effectively creating a long path length and high sensitivity while keeping the sample size small. Differential measurements allow for continuous correction of system baseline extinction losses. The CRDS sensor is used to measure GEM in ambient air and laboratory studies at 25 Hz, as well as to measure GEM fluxes using the eddy covariance (EC) flux measurement technique which is the first application of EC for GEM. With these measurements we aim to improve the quantification of dry deposition of GEM which is confounded by the bi-directional movement of GEM between surfaces and the atmosphere, to better measure fast concentration fluctuations from sources and sinks and during chemical transformations. Results of this study are published in Atmospheric Measurement Techniques.
Funding provided by NSF (Major Instrumentation)
March 2013. Response of soil mercury stocks to climate change
By Oleksandra Hararuk. Most of the mercury contained in an ecosystem resides in soils and is closely associated with soil carbon. Soil carbon is sensitive to changes in environmental factors, such as atmospheric CO₂, temperature and rainfall, all of which will change in the coming 100 years as projected by multiple earth system models. Apart from association with soil carbon mercury is associated with precipitation, as it is a major source of mercury deposition into an ecosystem. Using a present-day empirical relationship between mercury to carbon ratio and latitude, precipitation, and clay, we explored the sensitivity of soil mercury to climate change using a global earth system model to run the climate change scenarios. The results, published in Biogeosciences, indicate that soil mercury may increase by as much as 150% from the present-day values after 100 years of increasing CO₂, temperatures, and precipitation; or decrease up to 50% under increased CO₂, temperatures, and decreased precipitation.
Funding provided by EPA STAR
By Oleksandra Hararuk. Most of the mercury contained in an ecosystem resides in soils and is closely associated with soil carbon. Soil carbon is sensitive to changes in environmental factors, such as atmospheric CO₂, temperature and rainfall, all of which will change in the coming 100 years as projected by multiple earth system models. Apart from association with soil carbon mercury is associated with precipitation, as it is a major source of mercury deposition into an ecosystem. Using a present-day empirical relationship between mercury to carbon ratio and latitude, precipitation, and clay, we explored the sensitivity of soil mercury to climate change using a global earth system model to run the climate change scenarios. The results, published in Biogeosciences, indicate that soil mercury may increase by as much as 150% from the present-day values after 100 years of increasing CO₂, temperatures, and precipitation; or decrease up to 50% under increased CO₂, temperatures, and decreased precipitation.
Funding provided by EPA STAR

November 2012. Quantifying nutrient and mercury concentrations and loads in Lake Tahoe snowpack
By Chris Pearson. Lake Tahoe clarity has declined from 30 m readings in the 1960s to approximately 20 m today. Eutrophication from atmospheric and terrestrial nitrogen (N) and phosphorus (P) as well as light scattering by particulate inputs are the main causes of this decline. Along with N and P, mercury (Hg) is well-known pollutant due to its toxicity and ability to bio-accumulate in aquatic ecosystems. Previous studies have focused on direct deposition of nutrients and pollutants to the lake surface and little temporal or spatial data exists on the dynamics of atmospheric inputs to the basin’s snowpack. High variability in atmospheric deposition rates throughout the basin caused by complex terrain, precipitation gradients, and pollutant origins make lake level or single measurements insufficient to estimate total basin wide loads. My research will clarify sources of atmospheric N, P, and Hg input to the Lake Tahoe watershed through measurement and modeling of concentrations and loads within the basin’s snowpack. From the first snowfall until the end of melting, snowpack, covering 314 sq. miles of the basin, acts as a temporary storage for water and constituents that accumulate throughout winter and spring. With approximately 70 percent of precipitation falling as snow during the winter and spring months, atmospheric deposition contained in the basin’s snowpack is potentially a large driver of runoff and ground water pollutant levels. We directly measure N, P, and Hg concentrations and loads in Tahoe snowpack throughout the basin. These measurements include integrated snowpack samples, wet deposition collection, and storm-based surface samples to help quantify pollutant sources (i.e. in-basin/out-of-basin, wet/dry deposition) and volatilization loses. With these measurements, basin wide loads from winter and spring atmospheric deposition will be spatially mapped, quantified, sourced, and connected to precipitation levels to allow for improved protection and management of lake quality.
Funding provided by the DRI and the USGS (State Water Research Institute Program)
By Chris Pearson. Lake Tahoe clarity has declined from 30 m readings in the 1960s to approximately 20 m today. Eutrophication from atmospheric and terrestrial nitrogen (N) and phosphorus (P) as well as light scattering by particulate inputs are the main causes of this decline. Along with N and P, mercury (Hg) is well-known pollutant due to its toxicity and ability to bio-accumulate in aquatic ecosystems. Previous studies have focused on direct deposition of nutrients and pollutants to the lake surface and little temporal or spatial data exists on the dynamics of atmospheric inputs to the basin’s snowpack. High variability in atmospheric deposition rates throughout the basin caused by complex terrain, precipitation gradients, and pollutant origins make lake level or single measurements insufficient to estimate total basin wide loads. My research will clarify sources of atmospheric N, P, and Hg input to the Lake Tahoe watershed through measurement and modeling of concentrations and loads within the basin’s snowpack. From the first snowfall until the end of melting, snowpack, covering 314 sq. miles of the basin, acts as a temporary storage for water and constituents that accumulate throughout winter and spring. With approximately 70 percent of precipitation falling as snow during the winter and spring months, atmospheric deposition contained in the basin’s snowpack is potentially a large driver of runoff and ground water pollutant levels. We directly measure N, P, and Hg concentrations and loads in Tahoe snowpack throughout the basin. These measurements include integrated snowpack samples, wet deposition collection, and storm-based surface samples to help quantify pollutant sources (i.e. in-basin/out-of-basin, wet/dry deposition) and volatilization loses. With these measurements, basin wide loads from winter and spring atmospheric deposition will be spatially mapped, quantified, sourced, and connected to precipitation levels to allow for improved protection and management of lake quality.
Funding provided by the DRI and the USGS (State Water Research Institute Program)

March 2012. Development of a cold plate sampler to measure atmospheric mercury and volatile organic compounds
By Chris Moore. We have recently designed and initially tested a cold plate sampler (CPS) for daily collection of vapor-phase deposition (i.e., frost). The center piece of our CPS consists of a Peltier cooling element that cools an exposed stainless steel plate; warm air is dissipated away from the collection surface by a fan through a chimney on the warm side of the Peltier element, which prevents the fan from icing in cold conditions; and the cold plate is mounted upside-down in an enclosure for protection against the elements. As part of a NASA/JPL-funded study – "The Bromine, Ozone, and Mercury Experiment (BROMEX)" – in Barrow, Alaska, we performed initial testing of this CPS to force deposition of water vapor onto the stainless steel surface cooled far below the dew point. At the cold exposed surface, water vapor deposits from ambient air and generates an ice (frost) layer that can be sampled daily using trace-metal protocols. Our results showed a linear relationship (r2 = 0.32) between total Hg concentrations in collected ice deposition and atmospheric gaseous oxidized mercury (GOM) concentrations. While not perfect, the correlations do not account for environmental variables (such as wind speed and surface temperature) that are expected to influence GOM collection efficiency of the CPS (see below). Because GOM is the most water-soluble form of atmospheric Hg, we hypothesize that Hg accumulation may be due to dissolution of GOM in a quasi-liquid layer during formation of new ice. Although these initial tests were performed in the Arctic, we believe after further extensive testing that this technology may be deployed at almost any ambient temperature to act as surrogate surface measurement technique.
Funding provided by the DRI (Division of Atmospheric Sciences – EDGES)
By Chris Moore. We have recently designed and initially tested a cold plate sampler (CPS) for daily collection of vapor-phase deposition (i.e., frost). The center piece of our CPS consists of a Peltier cooling element that cools an exposed stainless steel plate; warm air is dissipated away from the collection surface by a fan through a chimney on the warm side of the Peltier element, which prevents the fan from icing in cold conditions; and the cold plate is mounted upside-down in an enclosure for protection against the elements. As part of a NASA/JPL-funded study – "The Bromine, Ozone, and Mercury Experiment (BROMEX)" – in Barrow, Alaska, we performed initial testing of this CPS to force deposition of water vapor onto the stainless steel surface cooled far below the dew point. At the cold exposed surface, water vapor deposits from ambient air and generates an ice (frost) layer that can be sampled daily using trace-metal protocols. Our results showed a linear relationship (r2 = 0.32) between total Hg concentrations in collected ice deposition and atmospheric gaseous oxidized mercury (GOM) concentrations. While not perfect, the correlations do not account for environmental variables (such as wind speed and surface temperature) that are expected to influence GOM collection efficiency of the CPS (see below). Because GOM is the most water-soluble form of atmospheric Hg, we hypothesize that Hg accumulation may be due to dissolution of GOM in a quasi-liquid layer during formation of new ice. Although these initial tests were performed in the Arctic, we believe after further extensive testing that this technology may be deployed at almost any ambient temperature to act as surrogate surface measurement technique.
Funding provided by the DRI (Division of Atmospheric Sciences – EDGES)
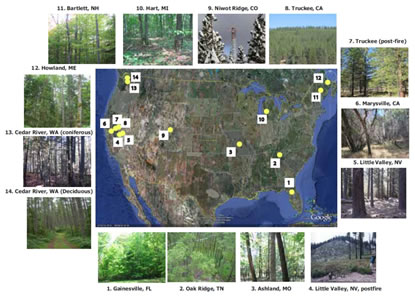
September 2011. Accumulation and fate processes of mercury in terrestrial ecosystems.
By Daniel Obrist. Terrestrial ecosystems serve as a key link between atmospheric deposition of Hg – the main pathway for Hg input in remote ecosystems – and mobilization to streams, lakes, and oceans where Hg pollution impacts wildlife and humans through consumption of Hg-laden fish. Soils account for more than 90% of Hg stored in terrestrial ecosystems, and top soil Hg pools (approximately 40 cm) are estimated to be as high as 15,230 metric tons in the U.S., or, if extrapolated globally, may account for more than 300,000 metric tons. This large pool size stems in part from natural, geologic sources but also is due to a large legacy of past anthropogenic pollution released across centuries that has accumulated in soils. Atmospheric Hg deposition is efficiently retained in upper soils, in particular bound in humus-rich layers. Due to the semi-volatile nature of elemental mercury (Hg0), however, terrestrial Hg also partially volatilizes into the atmosphere, a process accounting for up to one-third of total atmospheric Hg emissions. Our studies aim to characterize fate processes of mercury in soils, litter, and vegetation to understand the effects of large terrestrial Hg pools for future atmospheric mercury loads and mobilization to watersheds. This is done by field investigations and experimental laboratory studies to assess the behavior of mercury in soils, litter, and vegetation. More information in published articles: Environmental Science & Technology, Biogeosciences, and Science of the Total Environment.
Funding provided by EPA STAR
By Daniel Obrist. Terrestrial ecosystems serve as a key link between atmospheric deposition of Hg – the main pathway for Hg input in remote ecosystems – and mobilization to streams, lakes, and oceans where Hg pollution impacts wildlife and humans through consumption of Hg-laden fish. Soils account for more than 90% of Hg stored in terrestrial ecosystems, and top soil Hg pools (approximately 40 cm) are estimated to be as high as 15,230 metric tons in the U.S., or, if extrapolated globally, may account for more than 300,000 metric tons. This large pool size stems in part from natural, geologic sources but also is due to a large legacy of past anthropogenic pollution released across centuries that has accumulated in soils. Atmospheric Hg deposition is efficiently retained in upper soils, in particular bound in humus-rich layers. Due to the semi-volatile nature of elemental mercury (Hg0), however, terrestrial Hg also partially volatilizes into the atmosphere, a process accounting for up to one-third of total atmospheric Hg emissions. Our studies aim to characterize fate processes of mercury in soils, litter, and vegetation to understand the effects of large terrestrial Hg pools for future atmospheric mercury loads and mobilization to watersheds. This is done by field investigations and experimental laboratory studies to assess the behavior of mercury in soils, litter, and vegetation. More information in published articles: Environmental Science & Technology, Biogeosciences, and Science of the Total Environment.
Funding provided by EPA STAR

November 2010. Mercury in the Dead Sea
By Daniel Obrist. The atmosphere over the Dead Sea is laden with oxidized mercury. Some of the highest levels of oxidized mercury ever observed outside the polar regions exist there. The results published in Nature Geoscience. In the research, Daniel Obrist and colleagues at DRI and at Hebrew University in Isreal measured several periods of extremely high atmospheric oxidized mercury. Mercury exists in the atmosphere in an elemental and in an oxidized state. High levels of oxidized mercury are a concern because this form is deposited quickly in the environment after its formation. Observations of high naturally-occurring oxidized mercury levels had been limited to the polar atmosphere. There, oxidized mercury is formed during a process called atmospheric mercury depletion events: elemental mercury is converted to oxidized mercury, which is then readily deposited on surfaces. Now, we have found near-complete depletion of elemental mercury – and formation of some of the highest oxidized mercury levels ever seen – above the Dead Sea, a place where temperatures reach 45˚C. Such pronounced mercury depletion events were unexpected outside the frigid poles. High temperatures were thought to impede this chemical process. "Elemental mercury is somewhat resistant to oxidation, so it's been difficult to explain levels of oxidized mercury measured in the atmosphere outside polar regions," says Alex Pszenny, director of NSF's Atmospheric Chemistry Program, which funded the research. These new results provide an explanation. The mechanisms involved in the conversion of mercury above the Dead Sea appear similar, however, to those in polar regions: both start with halogens. Halogens, or halogen elements, are non-metal elements such as fluorine, chlorine, bromine and iodine. Observations and modeling results indicate that at the Dead Sea, the conversion of elemental mercury is driven by bromine. The new results show that bromine levels observed above oceans may be high enough to initiate mercury oxidation. We discovered that bromine can oxidize mercury in the mid-latitude atmosphere far from the poles. That points to an important role of bromine-induced mercury oxidation in mercury deposition over the world's oceans. What goes into the ocean may eventually wind up in its fish. And in those who eat them.
Funding provided by the DRI and the USGS (State Water Research Institute Program)
By Daniel Obrist. The atmosphere over the Dead Sea is laden with oxidized mercury. Some of the highest levels of oxidized mercury ever observed outside the polar regions exist there. The results published in Nature Geoscience. In the research, Daniel Obrist and colleagues at DRI and at Hebrew University in Isreal measured several periods of extremely high atmospheric oxidized mercury. Mercury exists in the atmosphere in an elemental and in an oxidized state. High levels of oxidized mercury are a concern because this form is deposited quickly in the environment after its formation. Observations of high naturally-occurring oxidized mercury levels had been limited to the polar atmosphere. There, oxidized mercury is formed during a process called atmospheric mercury depletion events: elemental mercury is converted to oxidized mercury, which is then readily deposited on surfaces. Now, we have found near-complete depletion of elemental mercury – and formation of some of the highest oxidized mercury levels ever seen – above the Dead Sea, a place where temperatures reach 45˚C. Such pronounced mercury depletion events were unexpected outside the frigid poles. High temperatures were thought to impede this chemical process. "Elemental mercury is somewhat resistant to oxidation, so it's been difficult to explain levels of oxidized mercury measured in the atmosphere outside polar regions," says Alex Pszenny, director of NSF's Atmospheric Chemistry Program, which funded the research. These new results provide an explanation. The mechanisms involved in the conversion of mercury above the Dead Sea appear similar, however, to those in polar regions: both start with halogens. Halogens, or halogen elements, are non-metal elements such as fluorine, chlorine, bromine and iodine. Observations and modeling results indicate that at the Dead Sea, the conversion of elemental mercury is driven by bromine. The new results show that bromine levels observed above oceans may be high enough to initiate mercury oxidation. We discovered that bromine can oxidize mercury in the mid-latitude atmosphere far from the poles. That points to an important role of bromine-induced mercury oxidation in mercury deposition over the world's oceans. What goes into the ocean may eventually wind up in its fish. And in those who eat them.
Funding provided by the DRI and the USGS (State Water Research Institute Program)
